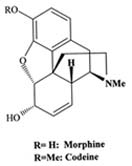
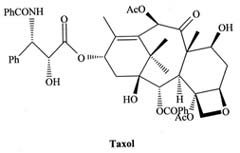
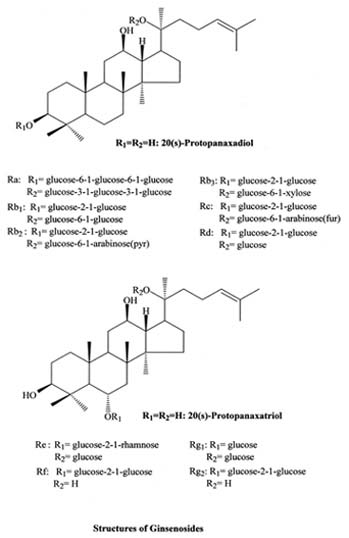
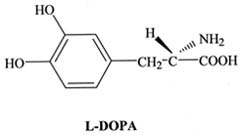
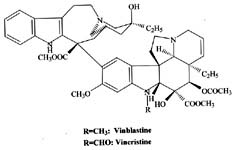
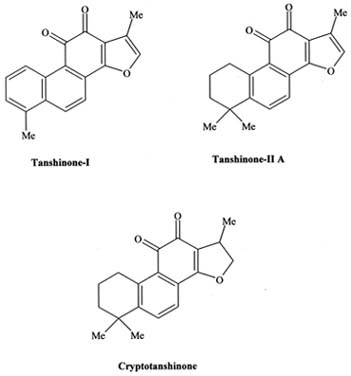
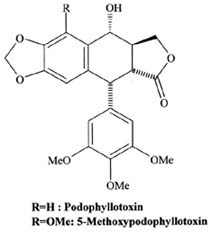
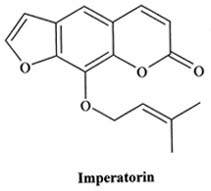
|
carboxylic acid and and amino acid feeding to cell cultures
of T. cuspidata. Biotechnol. Bioeng. 44: 967-971.
Franklin, C.I. and R.A. Dixon. 1994. Initiation and
maintenance of callus and cell suspension cultures. In R.A. Dixon and
R.A. Gonzales (eds.), Plant Cell Culture-A Practical Approach, 2nd
edn. IRL Press, Oxford, pp. 1-25.
Fujita, Y., Y. Hara, T. Ogino, and C. Suga. 1981.
Production of shikonin derivatives by cell suspension cultures of
Lithospermum erythrorhizon. Plant Cell Rep. 1: 59-60.
Fukui, H., M. Tani, and M. Tabata. 1990. Induction of
shikonin biosynthesis by endogenous polysaccharides in Lithospermum
erythrorhizon cell suspension cultures. Plant Cell Rep. 9:
73-76.
Funk, C., K. Gugler, and P. Brodelius. 1987. Increased
secondary product formation in plant cell suspension cultures after treatment
with a yeast carbohydrate preparation (elicitor). Phytochemistry 26:
401-405.
Furuya, T. 1988. Saponins (ginseng saponins). In
I.K. Vasil (ed.), Cell Cultures and Somatic Cell Genetics of Plants, Vol. 5.
Academic Press, San Diego, CA, pp. 213-234.
Furuya, T., A. Ikuta, and K. Syono. 1972. Alkaloids from
callus cultures of Papaver somniferum. Phytochemistry 11:
3041-3044.
Furuya, T., H. Kojima, K. Syono, T. Ishi, K. Uotani, and M.
Nishio. 1973. Isolation of saponin and sapogenins from callus tissue of
Panax ginseng. Chem. Pharm. Bull. 21: 98-101.
Furuya, T., T. Yoshikawa, Y. Orihara, and H. Oda. 1984.
Studies of the culture conditions for Panax ginseng cells in jar
fermentors. J. Nat. Prod. 47: 70-75.
Gamborg, O.L., R.A. Miller, and K. Ojima. 1968. Nutrient
requirements of suspension cultures of soyabean root cells. Exp. Cell Res.
50: 151-158.
Gantet, P. and J. Memelink. 2002. Transcription factors:
tools to engineer the production of pharmacologically active plant
metabolites. Trends Pharmacol. Sci. 23: 563-569.
Gerasimenko, I., Y. Sheludko, and J. Stockigt. 2001.
3-Oxo-rhazinilam: A new indole alkaloid from Rauwolfia serpentina x
Rhazya stricta hybrid plant cell cultures. J. Nat. Prod. 64:
114-116.
Giri, A. and M.L. Narasu. 2000. Transgenic hairy roots:
recent trends and applications. Biotechnol. Adv. 18: 1-22.
Goleniowski, M. and V.S. Trippi. 1999. Effect of growth
medium composition on psilostachyinolides and altamisine production. Plant
Cell Tiss. Org. Cult. 56: 215-218.
Guggenheim, M. 1913. Dioxyphenylalanin, eine neue
Aminosaure aus Vinca faba. Z. Physiol. Chem. 88: 276.
Hagimori, M., T. Matsumoto, and Y. Obi. 1982. Studies on
the production of Digitalis cardenolides by plant tissue culture. III.
Effects of nutrients on digitoxin formation by shoot-forming cultures of
Digitalis purpurea L. grown in liquid media. Plant Cell Physiol.
23(7): 1205-1211.
Hahn, J.J., C.A. Eschenlauer, H.M. Narrol, A.D. Somers, and
F. Srienc. 1997. Growth kinetics, nutrient uptake, and expression of the
Alcaligenes eutrophus poly(-hydroxybutarate) synthesis pathway in
transgenic maize cell suspension cultures. Biotechnol. Prog. 13(4):
347-354.
Hansen, G. and M.S. Wright. 1999. Recent advances in the
transformation of plants. Trends Plant Sci. 4: 226-231.
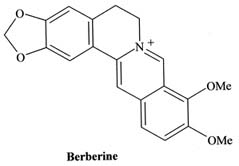
Hara, M., Y. Kobayashi, H. Fukui, and M. Tabata. 1991.
En
|
|
hancement of berberine production by spermidine in
Thalictrum minus cell suspension cultures. Plant Cell Rep. 10:
494-497.
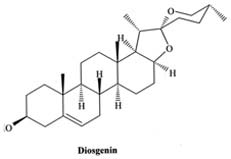
Heble, M. R., S. Narayanaswamy, and M.S. Chadha. 1967.
Diosgenin production and beta-sitosterol isolation from Solanum
xanthocarpum tissue cultures. Science 161: 1145.
Heble, M.R. and E. Staba. 1980. Steroid metabolism in
stationary phase cell suspensions of Dioscorea deltoidea. Planta Med.
Suppl., pp. 124-128.
Hiatt, A., R. Cafferkey, and K. Boedish. 1989. Production
of antibodies in transgenic plants. Nature 342: 76-78.
Holton, R.A., C. Somoza, H.B. Kim, F. Liang, R J. Biediger,
P. D. Boatman, M. Shindo, C.C. Smith, S. Kim, H. Nadizadeh, Y. Suzuki, C. Tao,
P. Vu, S. Tang, P. Zhang, K.K. Murthi, L.N. Gentile, and J.H. Liu. 1994. First
total synthesis of taxol. 1. Functionalization of the B ring. J. Am. Chem.
Soc. 116: 1597-1600.
Holden, R.R., M.A. Holden, and M.M. Yeoman. 1988. The
effects of fungal elicitation on secondary metabolism in cell cultures of
Capsicum frutescens. In R.J. Robins and M.J.C. Rhodes (eds.),
Manipulating secondary metabolism in culture, Cambridge University Press, pp.
67-72.
Hu, Z.B. and A.W. Alfermann. 1993. Diterpenoid production
in hairy root cultures of Salvia miltiorrhiza. Phytochemistry
32: 699-703.
Huang, K.C. 1993a. The pharmacology of Chinese herbs. CRC
Press, Boca Raton, FL, pp. 21-45.
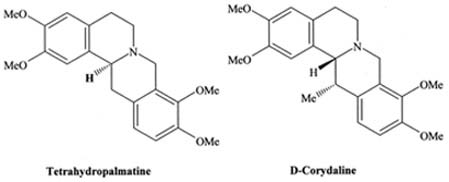
Huang, K.C. 1993b. Yan Hu Suo- The dried tuber of
Corydalis turtschaninovii Bess f. yanhusuo (Papaveraceae). In: The
pharmacology of Chinese herbs, CRC Press, Boca Raton, Florida, U.S.A. pp.
141-142.
Huang, W.W., C.C. Cheng, F.T. Yeh, and H.S. Tsay. 1993.
Tissue culture of Dioscorea doryophora HANCE 1. Callus organs and the
measurement of diosgenin content. Chin. Med. Coll. J. 2(2):
151-160.
Hwang, B., K.M. Ko, K.H. Hwang, S.J. Hwang, and Y.H. Kong.
1991. Production of saponin by hairy root cultures of ginseng (Panax
ginseng CA Mayer) transformed with Agrobacterium rhizogenes. Korean
J. Biotechnol. 34: 289-296.
Hyenga, A.G., J.A. Lucas, and P.M. Dewick, 1990. Production
of tumour-inhibitory lignans in callus cultures of Podophyllum
hexandrum. Plant Cell Rep. 9: 382-385.
Ikuta, A. and H. Itokawa. 1988. Alkaloids of tissue
cultures of Nandina domestica. Phytochemistry 27(7):
2143-2145.
Ishida, B.K. 1988. Improved diosgenin production in
Dioscorea deltoidea cell cultures by immobilization in polyurethane foam.
Plant Cell Rep. 7: 270-273.
Issell, B.F., A.R. Rudolph, and A.C. Louie. 1984. Etoposide
(VP-16-213): an overview. In B.F. Issell, F.M. Muggia, and S.K. Carter,
(eds.), Etoposide (VP-16-213)- Current status and new developments. Academic
Press Inc, Orlando, pp. 1-13.
Iwasa, K. and N. Takao. 1982. Formation of alkaloids in
Corydalis ophiocarpa callus cultures. Phytochemistry 21(3):
611-614.
Jain, M., A.K. Rathore, and P. Khanna. 1984. Influence of
kinetin and auxins on the growth and production of diosgenin by Costus
speciosus (Koen) Sm. callus derived from rhizome. Agri. Biol. Chem.
48: 529.
Jain, S.C. and S.L. Sahoo. 1981. Growth and production of
ste
|
|